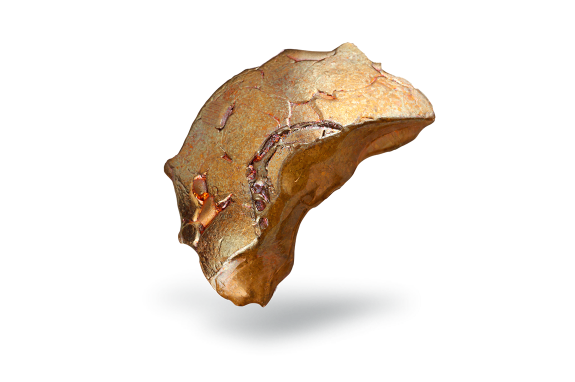
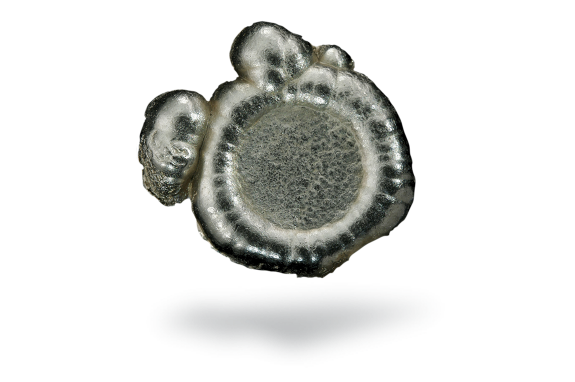
Tellurium
Whether you have a solar panel powering your home, drinking a glass of wine or driving around town in winter, you are more linked to tellurium than you might think. Tellurium has many applications, especially in photovoltaics and thermoelectrics, which are found in car seat heating, small fridges and wine coolers.
Applications
Tellurium is used mainly in alloys, mostly combined with copper and stainless steel in order to improve their machinability. Small additions of this metal, generally in the form of manganese telluride, improve the corrosion resistance as well as the strength and hardness of the alloy. This can be very beneficial in the manufacture of products like compact discs, copy machines and x-ray detectors.
Tellurium also plays a key role in solar panel technology. The semiconductor properties of tellurium, when alloyed with cadmium, creates Cd-Te that enables the conversion of sunlight into electricity in solar panels. Tellurium has also been used to vulcanize rubber and as a catalyst in oil refining.
As bismuth telluride Bi2Te3 and lead telluride PbTe, it has found certain applications in the electronic and thermoelectric fields.
Recycling
Tellurium is recovered from some anode slimes from the copper refining industry. It is also recycled from defective (process rejects) and end of life solar panels. Another recycling stream is production scrap from BiTe manufacturing.
Properties
Tellurium is a silvery, brittle metal with a typical metallic luster, usually obtained as a grey powder. In the molten state, it strongly corrodes metals such as iron and copper.
Tellurium is very rare in the Earth’s crust, but abundant in space. It is sometimes found in its natural form, but is more often found with gold or other metals in mineral forms. It is obtained commercially from the anode muds produced during the electrolytic refining of copper. These contain up to about 8% tellurium.
Tellurium also burns in air with a greenish flame to form tellurium dioxide.
History
Tellurium was discovered in gold-bearing ores in Romania by Müller von Reichenstein in 1782. Müller researched the ore for three years and proved it contained a new element. He published his findings but went largely unnoticed.
In 1796, he sent a sample to Martin Klaproth, who confirmed his discovery. Klaproth was able to produce a pure sample and in 1798 he decided to call it Tellurium, derived from the Latin Tellus meaning earth.